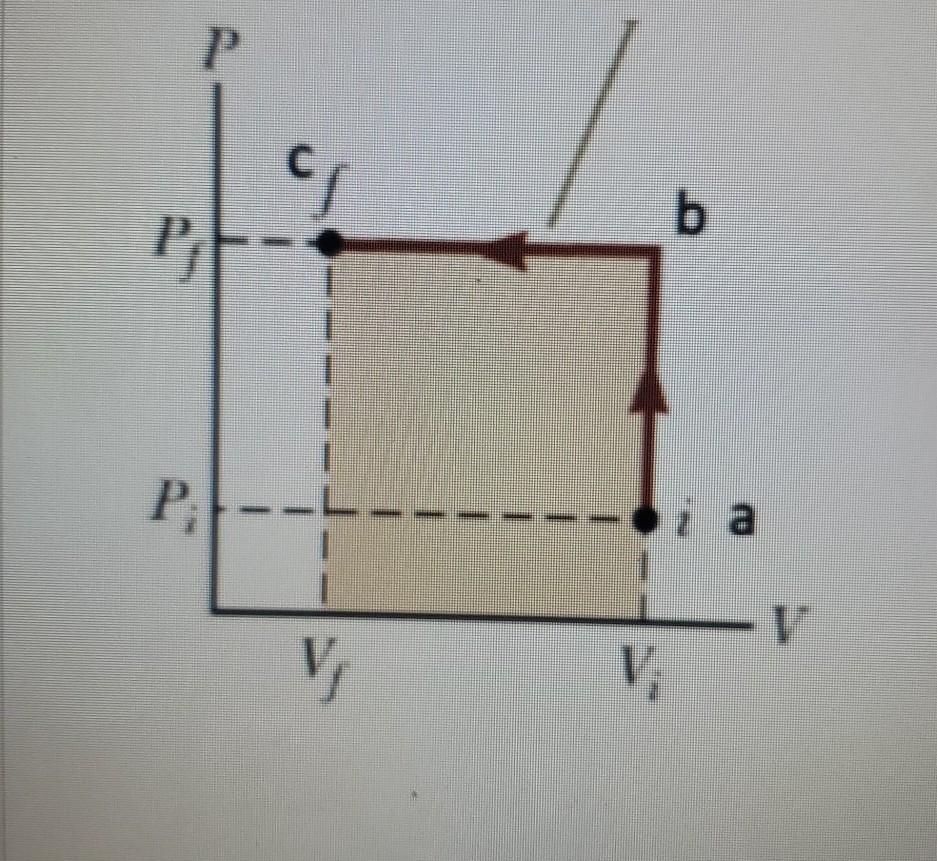
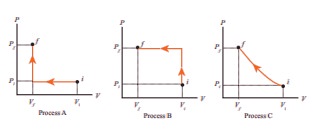
An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most. - Physics
By A Mystery Man Writer
Description
An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most.
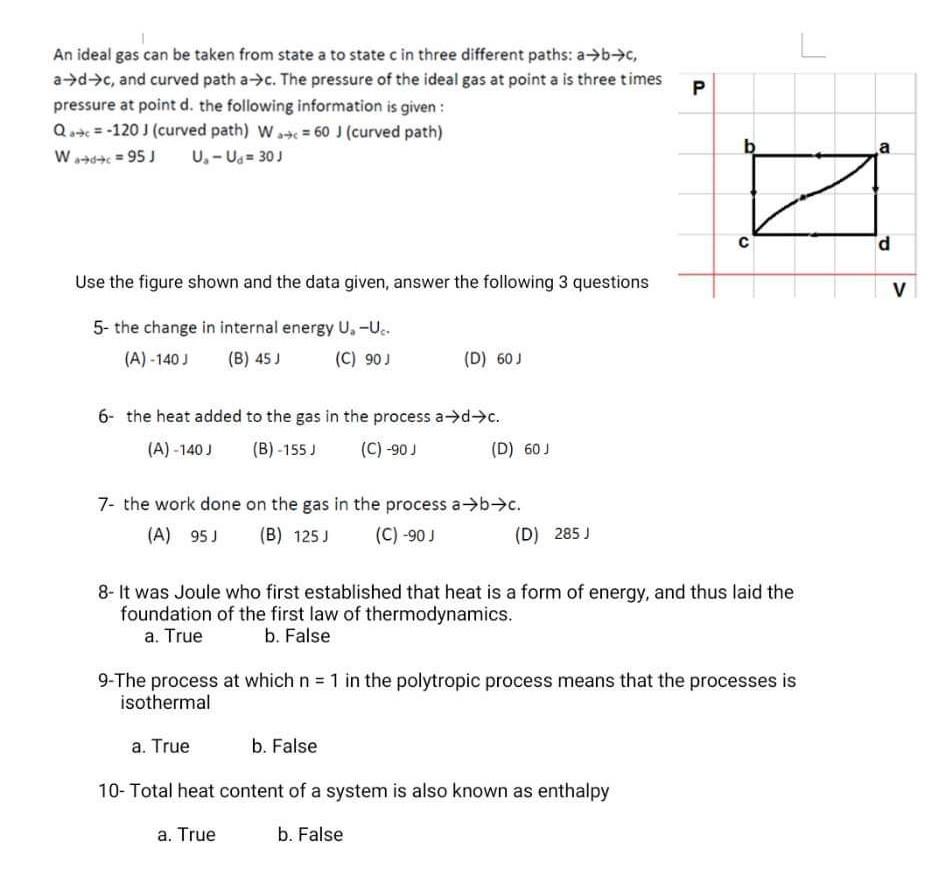
Solved An ideal gas can be taken from state a to state c in

5.8: Special Processes - Physics LibreTexts
Solved An ideal gas is taken from initial to final states

Ideal Gas Law Paper

An ideal gas goes from state A to state B via three different processes as indicated in the P-V diagram-If {Q}_{1}, {Q}_{2}, {Q}_{3} indicate the heat absorbed by the gas along the

Solved An ideal gas contained in a piston can change from
Five moles of an ideal gas are compressed isothermally from A to B, as the graph illustrates. What is the work involved if the temperature of the gas is 307 K? Be
UNIT VIII: Heat and Thermodynamics Online Objective Test

A gas expands from I to F along the three paths indicated in the figure. Calculate the work done on the gas along paths (a) IAF, (b) IF and (c) IBF.

Random, PDF, Gases

P-V diagram of an ideal gas is as shown in the given figure. Work done by the gas in the process

In figure, P-V curve of an ideal gas is given. During the process, the cumulative work done by t

JEE Advanced Physics-Waves and Thermodynamics [3 ed.] 9789354493140

One mole of an ideal gas is taken from state A to state B by three different processes a A C B, b A D B and c A E B as

Graphically show the total work done in an expansion when the state of an ideal gas is changed reversibly and isothermally from pi, Vi to pf, Vf With the help of p
from
per adult (price varies by group size)